Journal Description
Catalysts
Catalysts
is a peer-reviewed open access journal of catalysts and catalyzed reactions published monthly online by MDPI. The Romanian Catalysis Society (RCS) are partners of Catalysts journal and its members receive a discount on the article processing charge.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), Inspec, CAPlus / SciFinder, CAB Abstracts, and other databases.
- Journal Rank: JCR - Q2 (Chemistry, Physical) / CiteScore - Q1 (General Environmental Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.3 days after submission; acceptance to publication is undertaken in 2.7 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.9 (2022);
5-Year Impact Factor:
4.2 (2022)
Latest Articles
Glucose Isomerization to Fructose Catalyzed by MgZr Mixed Oxides in Aqueous Solution
Catalysts 2024, 14(5), 332; https://doi.org/10.3390/catal14050332 (registering DOI) - 18 May 2024
Abstract
The catalytic isomerization of glucose to fructose plays a pivotal role in the application of biomass as a feedstock for chemicals. Herein, we propose a facile solid-state-grinding strategy to construct ZrO2/MgO mixed oxides, which offered an excellent fructose yield of over
[...] Read more.
The catalytic isomerization of glucose to fructose plays a pivotal role in the application of biomass as a feedstock for chemicals. Herein, we propose a facile solid-state-grinding strategy to construct ZrO2/MgO mixed oxides, which offered an excellent fructose yield of over 34.55% and a high selectivity of 80.52% (80 °C, 2 h). The co-mingling of amphiphilic ZrO2 with MgO improved the unfavorable moderate/strongly basic site distribution on MgO, which can prohibit the side reactions during the reaction and enhance the fructose selectivity. Based on the catalyst characterizations, MgO was deposited on the ZrO2 surface by plugging the pores, and the addition of ZrO2 lessened the quantity of strongly basic sites of MgO. Additionally, the presence of ZrO2 largely enhanced the catalyst stability in comparison with pure MgO by recycling experiments.
Full article
(This article belongs to the Section Biomass Catalysis)
Open AccessEditorial
Nanotechnology in Catalysis, 2nd Edition
by
Maria J. Sabater
Catalysts 2024, 14(5), 331; https://doi.org/10.3390/catal14050331 - 17 May 2024
Abstract
Catalysis is considered a central field in nanoscience and nanotechnology, given that the use of nanoscale structures has played a central role in the development of nanomaterials such as catalysts (nanocatalysts) for decades [...]
Full article
(This article belongs to the Special Issue Nanotechnology in Catalysis, 2nd Edition)
Open AccessArticle
Investigating the Inhibitory Factors of Sucrose Hydrolysis in Sugar Beet Molasses with Yeast and Invertase
by
Mikael Sjölin, Maria Djärf, Mohamed Ismail, Herje Schagerlöf, Ola Wallberg, Rajni Hatti-Kaul and Mahmoud Sayed
Catalysts 2024, 14(5), 330; https://doi.org/10.3390/catal14050330 - 17 May 2024
Abstract
Sugar beet molasses is a low-value byproduct from the sugar industry. It contains significant amounts of sucrose (approx. 50% (w/w)), which can be used for many different applications, for example, as feedstock for the production of fuel (as ethanol)
[...] Read more.
Sugar beet molasses is a low-value byproduct from the sugar industry. It contains significant amounts of sucrose (approx. 50% (w/w)), which can be used for many different applications, for example, as feedstock for the production of fuel (as ethanol) and biobased chemicals such as 5-hydoxymethyl furfural (HMF). To produce platform chemicals, sucrose is hydrolyzed into its monomeric C6 sugars: glucose and fructose. When comparing the hydrolysis rates of molasses with a pure sucrose solution, the specific reaction rate is much slower (Qp/x,60min = 93 and 70 gprod L−1 h−1 gcell−1 for pure sucrose and crude molasses, respectively) at the same sucrose concentration (300 g/L) and process conditions. To clarify why molasses inhibits the enzymatic hydrolysis rate, the influence of its viscosity and inorganic and organic composition was investigated. Also, the effects of molasses and treated molasses on pure enzymes, invertase (from Saccharomyces cerevisiae, 0.05 mg/mL), compared with hydrolysis using whole cells of Baker’s yeast (3 mg/mL), were tested. The results indicate an inhibitory effect of potassium (Qp/x,60min = 76 gprod L−1 h−1 gcell−1), generally at high salt concentrations (Qp/x,60min = 67 gprod L−1 h−1 gcell−1), which could be correlated to the solution’s high salt concentrations and possibly the synergistic effects of different ions when applying concentrations that were four times that in the molasses. Also, the viscosity and sucrose purity seem to have an effect, where pure sucrose solutions and thick juice from the sugar mill yielded higher hydrolysis rates (Qp/x,60min = 97 gprod L−1 h−1 gcell−1) than molasses-type solutions with a higher viscosity (Qp/x,60min = 70–74 gprod L−1 h−1 gcell−1). Attempting to further understand the effects of different components on the invertase activity, an in silico investigation was performed, indicating that high salt concentrations affected the binding of sucrose to the active site of the enzyme, which can result in a lower reaction rate. This knowledge is important for future scale-up of the hydrolysis process, since reduced hydrolysis rates require larger volumes to provide a certain productivity, requiring larger process equipment and thereby higher investment costs.
Full article
(This article belongs to the Special Issue Applications of Catalytic Reactions in Promoting the Health of Organisms)
►▼
Show Figures

Figure 1
Open AccessArticle
Controllable Synthesis of Fe2O3/Nickel Cobaltite Electrocatalyst to Enhance Oxidation of Small Molecules
by
Fowzia S. Alamro, Shymaa S. Medany, Nada S. Al-Kadhi, Ayman M. Mostafa, Walaa F. Zaher, Hoda A. Ahmed and Mahmoud A. Hefnawy
Catalysts 2024, 14(5), 329; https://doi.org/10.3390/catal14050329 - 17 May 2024
Abstract
Nickel-based catalysts have been widely recognized as highly promising electrocatalysts for oxidation. Herein, we designed a catalyst surface based on iron oxide electrodeposited on NiCo2O4 spinel oxide. Nickel foam was used as a support for the prepared catalysts. The modified
[...] Read more.
Nickel-based catalysts have been widely recognized as highly promising electrocatalysts for oxidation. Herein, we designed a catalyst surface based on iron oxide electrodeposited on NiCo2O4 spinel oxide. Nickel foam was used as a support for the prepared catalysts. The modified surface was characterized by different techniques like electron microscopy and X-ray photon spectroscopy. The activity of the modified surface was investigated through the electrochemical oxidation of different organic molecules such as urea, ethanol, and ethylene glycol. Therefore, the modified Fe@ NiCo2O4/NF current in 1.0 M NaOH and 1.0 M fuel concentrations reached 31.4, 27.1, and 17.8 mA cm−2 for urea, ethanol, and ethylene glycol, respectively. Moreover, a range of kinetic characteristics parameters were computed, such as the diffusion coefficient, Tafel slope, and transfer coefficient. Chronoamperometry was employed to assess the electrode’s resistance to long-term oxidation. Consequently, the electrode’s activity exhibited a reduction ranging from 17% to 30% over a continuous oxidation period of 300 min.
Full article
(This article belongs to the Special Issue Recent Advances in Energy-Related Materials in Catalysts, 2nd Edition)
►▼
Show Figures

Graphical abstract
Open AccessReview
Recent Developments on CO2 Hydrogenation Performance over Structured Zeolites: A Review on Properties, Synthesis, and Characterization
by
Methene Briones Cutad, Mohammed J. Al-Marri and Anand Kumar
Catalysts 2024, 14(5), 328; https://doi.org/10.3390/catal14050328 - 17 May 2024
Abstract
This review focuses on an extensive synopsis of the recent improvements in CO2 hydrogenation over structured zeolites, including their properties, synthesis methods, and characterization. Key features such as bimodal mesoporous structures, surface oxygen vacancies, and the Si/Al ratio are explored for their
[...] Read more.
This review focuses on an extensive synopsis of the recent improvements in CO2 hydrogenation over structured zeolites, including their properties, synthesis methods, and characterization. Key features such as bimodal mesoporous structures, surface oxygen vacancies, and the Si/Al ratio are explored for their roles in enhancing catalytic activity. Additionally, the impact of porosity, thermal stability, and structural integrity on the performance of zeolites, as well as their interactions with electrical and plasma environments, are discussed in detail. The synthesis of structured zeolites is analyzed by comparing the advantages and limitations of bottom-up methods, including hard templating, soft templating, and non-templating approaches, to top-down methods, such as dealumination, desilication, and recrystallization. The review addresses the challenges associated with these synthesis techniques, such as pore-induced diffusion limitations, morphological constraints, and maintaining crystal integrity, highlighting the need for innovative solutions and optimization strategies. Advanced characterization techniques are emphasized as essential for understanding the catalytic mechanisms and dynamic behaviors of zeolites, thereby facilitating further research into their efficient and effective use. The study concludes by underscoring the importance of continued research to refine synthesis and characterization methods, which is crucial for optimizing catalytic activity in CO2 hydrogenation. This effort is important for achieving selective catalysis and is paramount to the global initiative to reduce carbon emissions and address climate change.
Full article
(This article belongs to the Special Issue Applications of Heterogeneous Catalysts in Green Chemistry)
►▼
Show Figures

Graphical abstract
Open AccessFeature PaperArticle
Study of Oxygen Reduction Reaction on Polycrystalline Rhodium in Acidic and Alkaline Media
by
Jelena Golubović, Miroslava Varničić and Svetlana Štrbac
Catalysts 2024, 14(5), 327; https://doi.org/10.3390/catal14050327 - 16 May 2024
Abstract
This study examines the kinetics and mechanism of the oxygen reduction reaction (ORR) on a polycrystalline rhodium electrode (Rh(poly)) in acidic and alkaline media, using rotating disc electrode measurements. This study found that the ORR activity of the Rh(poly) electrode decreases in the
[...] Read more.
This study examines the kinetics and mechanism of the oxygen reduction reaction (ORR) on a polycrystalline rhodium electrode (Rh(poly)) in acidic and alkaline media, using rotating disc electrode measurements. This study found that the ORR activity of the Rh(poly) electrode decreases in the order of 0.1 M NaOH > 0.1 M HClO4 > 0.05 M H2SO4 concerning the half-wave potentials. The Tafel slopes for ORR on Rh(poly) in the cathodic direction are 60 and 120 mV dec−1 at low and high overpotentials, respectively, in perchloric acid and alkaline solutions. However, strongly adsorbed sulfate anions hinder the ORR on Rh(poly) in sulfuric acid, leading to higher Tafel slopes. The highest ORR activity of Rh(poly) in an alkaline media suggests the promoting role of the specifically adsorbed OH− anions and RhOH. In all cases, ORR on Rh(poly) proceeds through the 4e-series reaction pathway.
Full article
(This article belongs to the Special Issue Exploring the Mechanisms and Kinetics of Electrocatalytic Reactions)
Open AccessArticle
Optimization of Desulfurization Process via Choline Phosphotungstate Coupled with Persulfate Using Response Surface Methodology
by
Yinke Zhang and Hang Xu
Catalysts 2024, 14(5), 326; https://doi.org/10.3390/catal14050326 - 16 May 2024
Abstract
Using a simple acid-base neutralization method, a Ch-PW solid catalyst was synthesized by mixing choline hydroxide (ChOH) and phosphotungstic acid (HPW) at a 2:1 molar ratio in an aqueous solution. This catalyst was combined with a 20 wt.% potassium peroxymonosulfate (PMS) solution, using
[...] Read more.
Using a simple acid-base neutralization method, a Ch-PW solid catalyst was synthesized by mixing choline hydroxide (ChOH) and phosphotungstic acid (HPW) at a 2:1 molar ratio in an aqueous solution. This catalyst was combined with a 20 wt.% potassium peroxymonosulfate (PMS) solution, using acetonitrile (ACN) as the extraction solvent to create an extraction catalytic oxidative desulfurization system. The optimal desulfurization conditions were determined through response surface methodology, targeting the highest desulfurization rate: 0.99 g of Ch-PW, 1.07 g of PMS, 2.5 g of extraction solvent, at a temperature of 50.48 °C. The predicted desulfurization rate was 90.79%, compared to an experimental rate of 93.64%, with a deviation of 3.04%. A quadratic model correlating the desulfurization rate with the four conditions was developed and validated using ANOVA, which also quantified the impact of each factor on the desulfurization rate: PMS > ACN > Ch-PW > temperature. GC-MS analysis identified the main oxidation product as DBTO2, and the mechanism of desulfurization in this system was further explored.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures

Figure 1
Open AccessArticle
Nano-Sheets of CsNiVF6 Pyrochlore Electrocatalyst for Enhanced Urea Oxidation and Hydrogen Green Production Reactions
by
Mohamed A. Ghanem, Abdullah M. Al-Mayouf, Khalaf A. Alfudhayli and Mohamed O. Abdelkader
Catalysts 2024, 14(5), 325; https://doi.org/10.3390/catal14050325 - 16 May 2024
Abstract
This study presents the successful synthesis of a cesium–nickel–vanadium fluoride (CsNiVF6) pyrochlore nano-sheet catalyst via solid-phase synthesis and its electrochemical performance in green hydrogen production through urea electrolysis in alkaline media. The physicochemical characterizations revealed that the CsNiVF6 exhibits a
[...] Read more.
This study presents the successful synthesis of a cesium–nickel–vanadium fluoride (CsNiVF6) pyrochlore nano-sheet catalyst via solid-phase synthesis and its electrochemical performance in green hydrogen production through urea electrolysis in alkaline media. The physicochemical characterizations revealed that the CsNiVF6 exhibits a pyrochlore-type structure consisting of a disordered cubic corner-shared (Ni, V)F6 octahedra structure and nano-sheet morphology with a thickness ranging from 10 to 20 nm. Using the CsNiVF6 catalyst, the electrochemical analysis, conducted through cyclic voltammetry, demonstrates a current mass activity of ~1500 mA mg−1, recorded at 1.8 V vs. RHE, along with low-resistance (3.25 ohm) charge transfer and good long-term stability for 0.33 M urea oxidation in an alkaline solution. Moreover, the volumetric hydrogen production rate at the cathode (bare nickel foam) is increased from 12.25 to 39.15 µmol/min upon the addition of 0.33 M urea to a 1.0 KOH solution and at a bias potential of 2.0 V. The addition of urea to the electrolyte solution enhances hydrogen production at the cathode, especially at lower voltages, surpassing the volumes produced in pure 1.0 M KOH solution. This utilization of a CsNiVF6 pyrochlore nano-sheet catalyst and renewable urea as a feedstock contributes to the development of a green and sustainable hydrogen economy. Overall, this research underscores the potential use of CsNiVF6 as a cost-effective nickel-based pyrochlore electrocatalyst for advancing renewable and sustainable urea electrolysis processes toward green hydrogen production.
Full article
(This article belongs to the Special Issue Electrocatalysis for Hydrogen/Oxygen Evolution Reactions)
►▼
Show Figures

Figure 1
Open AccessArticle
One-Step Synthesis of High-Efficiency Oxygen Evolution Reaction Catalyst FeSx(Y/MB) with High Temperature Resistance and Strong Alkali
by
Jing Wang, Lingling Feng, Zikang Zhao, Yan Wang, Ying Zhang, Shan Song, Shengwei Sun, Junshuang Zhou and Faming Gao
Catalysts 2024, 14(5), 324; https://doi.org/10.3390/catal14050324 - 16 May 2024
Abstract
Given the energy crisis and escalating environmental pollution, the imperative for developing clean new energy is evident. Hydrogen has garnered significant attention owing to its clean properties, high energy density, and ease of storage and transportation. This study synthesized four types of catalysts—FeS(DI/MB),
[...] Read more.
Given the energy crisis and escalating environmental pollution, the imperative for developing clean new energy is evident. Hydrogen has garnered significant attention owing to its clean properties, high energy density, and ease of storage and transportation. This study synthesized four types of catalysts—FeS(DI/MB), FeS(ET/MB), Fe(DI/MB), and Fe(ET/MB)—using two distinct solution systems: DI/MB and ET/MB. The FeS(DI/MB) catalyst, synthesized using the layered solution system (DI/MB), demonstrates a uniformly distributed and dense nanosheet structure, exhibiting excellent resistance to strong bases and superior catalytic properties. The FeS(DI/MB) electrode showed OER overpotentials of 460 mV and 318 mV in 1 M and 6 M, respectively, at current densities of up to 500 mA cm−2. Under industrial electrolysis test conditions, the FeS(DI/MB) electrode required only 262 mV to achieve a current density of 500 mA cm−2, operating in a high-temperature, strong alkaline environment of 6 M at 60 °C. Furthermore, the FeS(DI/MB) electrode exhibited excellent OER catalytic activity and stability, as evidenced by a 60 h stability test These findings provide valuable insights into the preparation of iron nickel sulfide-based catalysts, and further in-depth and comprehensive exploration is anticipated to yield the excellent catalytic performance of these catalysts in the realm of electrolytic water hydrogen production.
Full article
(This article belongs to the Special Issue Hierarchically Catalysts for Water Splitting and Selective Hydrogenation)
►▼
Show Figures

Figure 1
Open AccessArticle
Lithium Polysulfide Catalytic Mechanism of AlN/InN Heterojunction by First-Principles Calculation
by
Lingfeng Ye, Jin Wang, Zhiping Lin, Huafeng Dong and Fugen Wu
Catalysts 2024, 14(5), 323; https://doi.org/10.3390/catal14050323 - 14 May 2024
Abstract
To solve the shuttling effect and transformations of LiPSs in lithium–sulfur batteries, heterostructures have been designed to immobilize LiPSs and boost their reversible conversions. In this paper, we have constructed AlN/InN heterojunctions with AlN with a wide band gap and InN with a
[...] Read more.
To solve the shuttling effect and transformations of LiPSs in lithium–sulfur batteries, heterostructures have been designed to immobilize LiPSs and boost their reversible conversions. In this paper, we have constructed AlN/InN heterojunctions with AlN with a wide band gap and InN with a narrow band gap. The heterojunctions show metallic properties, which are primarily composed of 2s, 2p N atoms and 5s, 5p In atoms. InN has relatively higher adsorptivity for LiPSs than AlN. Reaction profiles show that on the surface of AlN, there is a lower rate-limiting step than on that of InN, from S8 to Li2S6, and a higher rate-limiting step from Li2S4 to Li2S2, which is more favorable for InN during the reduction from Li2S4 to Li2S2. The heterojunction can realize the synergistic reaction of trapping–diffusion–conversion for LiPSs, in which AlN traps large Li2S8 and Li2S6, the heterojunction causes the diffusion of Li2S4, and InN completes the conversion of Li2S4 to Li2S.
Full article
(This article belongs to the Section Computational Catalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Performance Evaluation and Durability Analysis of NiFeCoOx Catalysts for Alkaline Water Electrolysis in Anion Exchange Membrane Electrolyzers
by
Khaja Wahab Ahmed and Michael Fowler
Catalysts 2024, 14(5), 322; https://doi.org/10.3390/catal14050322 - 14 May 2024
Abstract
This study examines the catalytic activity of NiFeCoOx catalysts for anion exchange membrane (AEM) water electrolysis. The catalysts were synthesized with a Ni to Co ratio of 2:1 and Fe content ranges from 2.5 to 12.5 wt%. The catalysts were characterized using
[...] Read more.
This study examines the catalytic activity of NiFeCoOx catalysts for anion exchange membrane (AEM) water electrolysis. The catalysts were synthesized with a Ni to Co ratio of 2:1 and Fe content ranges from 2.5 to 12.5 wt%. The catalysts were characterized using scanning electron microscopy (SEM) and X-ray diffraction (XRD) techniques. The catalytic activity of the NiFeCoOx catalysts was evaluated through linear sweep voltammetry (LSV) and chronoamperometry (CA) experiments for the oxygen evolution reaction (OER). The catalyst with 5% Fe content exhibited the highest catalytic activity, achieving an overpotential of 228 mV at a current density of 10 mA cm−2. Long-term catalyst testing for the OER at 50 mA cm−2 showed stable electrolysis operation for 100 h. The catalyst was further analyzed in an AEM water electrolyzer in a single-cell test, and the NiFeCoOx catalyst with 5% Fe at the anode demonstrated the highest current densities of 1516 mA cm−2 and 1620 mA cm−2 at 55 °C and 70 °C at 2.1 V. The maximum current density of 1880 mA cm−2 was achieved at 2.2 V and 70 °C. The Nyquist plot analysis of electrolysis at 55 °C showed that the NiFeCoOx catalyst with 5% Fe had lower activation resistance compared with the other Fe loadings, indicating enhanced performance. The durability test was performed for 8 h, showing stable AEM water electrolysis with minimum degradation. An overall cell efficiency of 70.5% was achieved for the operation carried out at a higher current density of 0.8 A cm−2.
Full article
(This article belongs to the Section Electrocatalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Facile Asymmetric Syntheses of Non-Natural Amino Acid (S)-Cyclopropylglycine by the Developed NADH-Driven Biocatalytic System
by
Qian Tang, Shanshan Li, Liping Zhou, Lili Sun, Juan Xin and Wei Li
Catalysts 2024, 14(5), 321; https://doi.org/10.3390/catal14050321 - 13 May 2024
Abstract
A self-sufficient bifunctional enzyme integrating reductive amination and coenzyme regeneration activities was developed and successfully employed to synthesize (S)-cyclopropylglycine with an improved reaction rate 2.1-fold over the native enzymes and a short bioconversion period of 6 h at a high substrate
[...] Read more.
A self-sufficient bifunctional enzyme integrating reductive amination and coenzyme regeneration activities was developed and successfully employed to synthesize (S)-cyclopropylglycine with an improved reaction rate 2.1-fold over the native enzymes and a short bioconversion period of 6 h at a high substrate concentration of 120 g·L−1 and space–time yield of (S)-cyclopropylglycine up to 377.3 g·L−1·d−1, higher than that of any previously reported data. Additionally, (S)-cyclopropylglycine could be continuously synthesized for 90 h with the enzymes packed in a dialysis tube, providing 634.6 g of (S)-cyclopropylglycine with >99.5% ee and over 95% conversion yield up to 12 changes. These results confirmed that the newly developed NADH-driven biocatalytic system could be utilized as a self-sufficient biocatalyst for industrial application in the synthesis of (S)-cyclopropylglycine, which provides a chiral center and cyclopropyl fragment for the frequent synthesis of preclinical/clinical drug molecules.
Full article
(This article belongs to the Section Biocatalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Liquid–Liquid Equilibrium Behavior of Ternary Systems Comprising Biodiesel + Glycerol and Triglyceride + Methanol: Experimental Data and Modeling
by
Lingmei Yang, Shiyou Xing, Xianbin Teng, Rukuan Liu, Zhongming Wang, Baining Lin, Pengmei Lv, Akram Ali Nasser Mansoor Al-Haimi, Fatma Yehia and Wen Luo
Catalysts 2024, 14(5), 320; https://doi.org/10.3390/catal14050320 - 12 May 2024
Abstract
Having a comprehensive knowledge of phase equilibrium is advantageous for industrial simulation and design of chemical processes. For further acquisition of primary data to facilitate the separation and purification of waste oil biodiesel systems, a liquid–liquid equilibrium (LLE) tank is deployed for the
[...] Read more.
Having a comprehensive knowledge of phase equilibrium is advantageous for industrial simulation and design of chemical processes. For further acquisition of primary data to facilitate the separation and purification of waste oil biodiesel systems, a liquid–liquid equilibrium (LLE) tank is deployed for the ternary system of waste oil biodiesel + methanol + glycerin, thereby enhancing the precision and efficiency of the process. The phase equilibrium system was constructed under the influence of atmospheric pressure at precise temperatures of 303.15 K, 313.15 K, and 323.15 K. The equilibrium components of each substance were analyzed by employing high-temperature gas chromatography, a sophisticated analytical method that enables the identification and quantification of individual components of a sample. Moreover, the ternary liquid–liquid equilibrium data were correlated by implementing the NRTL and UNIQUAC activity coefficient models. Subsequently, the binary interaction parameters of the ternary system were derived by conducting regression analysis. The experimental data demonstrated that the presence of lower methanol content in the system resulted in nearly immiscible biodiesel and glycerol phases, which ultimately facilitated the separation of biodiesel and glycerol. Conversely, with the increase in methanol content, the mutual solubility of biodiesel and glycerol was observed to increase gradually. The results showed that the calculated values of the NRTL and UNIQUAC models aligned well with the experimental values. The root-mean-square deviations of the NRTL and UNIQUAC models at 313.15 K were 2.76% and 3.56%, respectively.
Full article
(This article belongs to the Section Biomass Catalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Methane Combustion Kinetics over Palladium-Based Catalysts: Review and Modelling Guidelines
by
Roshni Sajiv Kumar, Joseph P. Mmbaga, Natalia Semagina and Robert E. Hayes
Catalysts 2024, 14(5), 319; https://doi.org/10.3390/catal14050319 - 11 May 2024
Abstract
Fugitive methane emissions account for a significant proportion of greenhouse gas emissions, and their elimination by catalytic combustion is a relatively easy way to reduce global warming. New and novel reactor designs are being considered for this purpose, but their correct and efficient
[...] Read more.
Fugitive methane emissions account for a significant proportion of greenhouse gas emissions, and their elimination by catalytic combustion is a relatively easy way to reduce global warming. New and novel reactor designs are being considered for this purpose, but their correct and efficient design requires kinetic rate expressions. This paper provides a comprehensive review of the current state of the art regarding kinetic models for precious metal catalysts used for the catalytic combustion of lean methane mixtures. The primary emphasis is on relatively low-temperature operation at atmospheric pressure, conditions that are prevalent in the catalytic destruction of low concentrations of methane in emission streams. In addition to a comprehensive literature search, we illustrate a detailed example of the methodology required to determine an appropriate kinetic model and the constants therein. From the wide body of literature, it is seen that the development of a kinetic model is not necessarily a trivial matter, and it is difficult to generalize. The model, especially the dependence on the water concentration, is a function of not only the active ingredients but also the nature of the support. Kinetic modelling is performed for six catalysts, one commercial and five that were manufactured in our laboratory, for illustration purposes.
Full article
(This article belongs to the Section Computational Catalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Direct Synthesis of Calcium Lactate through the Reaction of Glycerol with Calcium Hydroxide Catalyzed by Bimetallic AuCu/SiO2 Nanocatalysts
by
Changqing Li, Xinyue Cui, Aili Wang, Hengbo Yin, Yuting Li, Qiao Lin and Junjie Guo
Catalysts 2024, 14(5), 318; https://doi.org/10.3390/catal14050318 - 11 May 2024
Abstract
Bimetallic AuCu/SiO2 nanosized catalysts were prepared using the wet chemical reduction technique. From among Au0.1–1.5Cu10/SiO2 catalysts, the Au0.5Cu10/SiO2 catalyst gave the highest yield of calcium lactate of 87% at a glycerol conversion
[...] Read more.
Bimetallic AuCu/SiO2 nanosized catalysts were prepared using the wet chemical reduction technique. From among Au0.1–1.5Cu10/SiO2 catalysts, the Au0.5Cu10/SiO2 catalyst gave the highest yield of calcium lactate of 87% at a glycerol conversion of 96% when the reaction of glycerol with calcium hydroxide at a mole ratio of calcium hydroxide to glycerol of 0.8:1 was conducted under an anaerobic atmosphere at 200 °C for 2 h. The interactions between metallic Au0 and Cu0 nanoparticles facilitate calcium lactate formation. The simulation of glycerol consumption rate with an empirical power-function reaction kinetics equation yielded a reaction activation energy of 44.3 kJ∙mol−1, revealing that the catalytic reaction of glycerol with calcium hydroxide to calcium lactate can be conducted by overcoming a mild energy barrier. The synthesis of calcium lactate through the catalytic reaction of glycerol with calcium hydroxide on a bimetallic AuCu/SiO2 nanosized catalyst under a safe anaerobic atmosphere is an alternative to the conventional calcium lactate production technique through the reaction of expensive lactic acid with calcium hydroxide.
Full article
(This article belongs to the Special Issue Heterogeneous Catalysis for Environmentally Compatible Reactions and Processes)
►▼
Show Figures

Graphical abstract
Open AccessReview
A Comprehensive Review of Fine Chemical Production Using Metal-Modified and Acidic Microporous and Mesoporous Catalytic Materials
by
Joseph Lantos, Narendra Kumar and Basudeb Saha
Catalysts 2024, 14(5), 317; https://doi.org/10.3390/catal14050317 - 10 May 2024
Abstract
Fine chemicals are produced in small annual volume batch processes (often <10,000 tonnes per year), with a high associated price (usually >USD 10/kg). As a result of their usage in the production of speciality chemicals, in areas including agrochemicals, fragrances, and pharmaceuticals, the
[...] Read more.
Fine chemicals are produced in small annual volume batch processes (often <10,000 tonnes per year), with a high associated price (usually >USD 10/kg). As a result of their usage in the production of speciality chemicals, in areas including agrochemicals, fragrances, and pharmaceuticals, the need for them will remain high for the foreseeable future. This review article assesses current methods used to produce fine chemicals with heterogeneous catalysts, including both well-established and newer experimental methods. A wide range of methods, utilising microporous and mesoporous catalysts, has been explored, including their preparation and modification before use in industry. Their potential drawbacks and benefits have been analysed, with their feasibility compared to newer, recently emerging catalysts. The field of heterogeneous catalysis for fine chemical production is a dynamic and ever-changing area of research. This deeper insight into catalytic behaviour and material properties will produce more efficient, selective, and sustainable processes in the fine chemical industry. The findings from this article will provide an excellent foundation for further exploration and a critical review in the field of fine chemical production using micro- and mesoporous heterogeneous catalysts.
Full article
(This article belongs to the Special Issue Microporous and Mesoporous Materials for Catalytic Applications)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Promoter Impact on 5Ni/SAPO-5 Catalyst for H2 Production via Methane Partial Oxidation
by
Abdulaziz Al-Anazi, Omer Bellahwel, Kavitha C., Jehad Abu-Dahrieh, Ahmed A. Ibrahim, S. Santhosh, Ahmed E. Abasaeed, Anis H. Fakeeha and Ahmed S. Al-Fatesh
Catalysts 2024, 14(5), 316; https://doi.org/10.3390/catal14050316 - 10 May 2024
Abstract
Compared to steam reforming techniques, partial oxidation of methane (POM) is a promising technology to improve the efficiency of synthesizing syngas, which is a mixture of CO and H2. In this study, partial oxidation of methane (POM) was used to create
[...] Read more.
Compared to steam reforming techniques, partial oxidation of methane (POM) is a promising technology to improve the efficiency of synthesizing syngas, which is a mixture of CO and H2. In this study, partial oxidation of methane (POM) was used to create syngas, a combination of CO and H2, using the SAPO-5-supported Ni catalysts. Using the wetness impregnation process, laboratory-synthesized Ni promoted with Sr, Ce, and Cu was used to modify the SAPO-5 support. The characterization results demonstrated that Ni is appropriate for the POM due to its crystalline structure, improved metal support contact, and increased thermal stability with Sr, Ce, and Cu promoters. During POM at 600 °C, the synthesized 5Ni+1Sr/SAPO-5 catalyst sustained stability for 240 min on stream. While keeping the reactants stoichiometric ratio of (CH4:O2 = 2:1), the addition of Sr promoter and active metal Ni to the SAPO-5 increased the CH4 conversion from 41.13% to 49.11% and improved the H2/CO ratio of 3.33. SAPO-5-supported 5Ni+1Sr catalysts have great potential for industrial catalysis owing to their unique combination of several oxides. This composition not only boosts the catalyst’s activity but also promotes favorable physiochemical properties, resulting in improved production of syngas. Syngas is a valuable intermediate in various industrial processes.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures
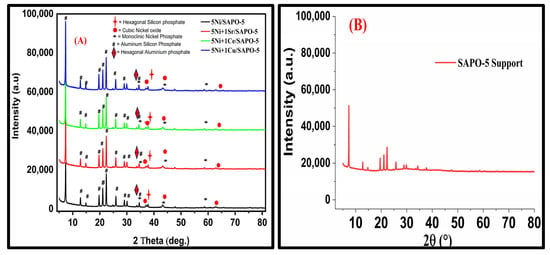
Figure 1
Open AccessArticle
Au Nanoparticles Supported on Hydrotalcite-Based MMgAlOx (M=Cu, Ni, and Co) Composite: Influence of Dopants on the Catalytic Activity for Semi-Hydrogenation of C2H2
by
Xun Sun, Wenrui Lv, Yanan Cheng, Huijuan Su, Libo Sun, Lijun Zhao, Zifan Wang and Caixia Qi
Catalysts 2024, 14(5), 315; https://doi.org/10.3390/catal14050315 - 10 May 2024
Abstract
Semi-hydrogenation of acetylene to ethylene over metal oxide-supported Au nanoparticles is an interesting topic. Here, a hydrotalcite-based MMgAlOx (M=Cu, Ni, and Co) composite oxide was exploited by introducing different Cu, Ni, and Co dopants with unique properties, and then used as support
[...] Read more.
Semi-hydrogenation of acetylene to ethylene over metal oxide-supported Au nanoparticles is an interesting topic. Here, a hydrotalcite-based MMgAlOx (M=Cu, Ni, and Co) composite oxide was exploited by introducing different Cu, Ni, and Co dopants with unique properties, and then used as support to obtain Au/MMgAlOx catalysts via a modified deposition–precipitation method. XRD, BET, ICP-OES, TEM, Raman, XPS, and TPD were employed to investigate their physic-chemical properties and catalytic performances for the semi-hydrogenation of acetylene to ethylene. Generally, the catalytic activity of the Cu-modified Au/CuMgAlOx catalyst was higher than that of the other modified catalysts. The TOR for Au/CuMgAlOx was 0.0598 h−1, which was 30 times higher than that of Au/MgAl2O4. The SEM and XRD results showed no significant difference in structure or morphology after introducing the dopants. These dopants had an unfavorable effect on the Au particle size, as confirmed by the TEM studies. Accordingly, the effects on catalytic performance of the M dopant of the obtained Au/MMgAlOx catalyst were improved. Results of Raman, NH3-TPD, and CO2-TPD confirmed that the Au/CuMgAlOx catalyst had more basic sites, which is beneficial for less coking on the catalyst surface after the reaction. XPS analysis showed that gold nanoparticles exhibited a partially oxidized state at the edges and surfaces of CuMgAlOx. Besides an increased proportion of basic sites on Au/CuMgAlOx catalysts, the charge transfer from nanogold to the Cu-doped matrix support probably played a positive role in the selective hydrogenation of acetylene. The stability and deactivation of Au/CuMgAlOx catalysts were also discussed and a possible reaction mechanism was proposed.
Full article
(This article belongs to the Special Issue Nanomaterials in Catalysis: Design, Characterization and Applications)
►▼
Show Figures

Figure 1
Open AccessArticle
Metal–Organic Framework Fe-BTC as Heterogeneous Catalyst for Electro-Fenton Treatment of Tetracycline
by
Taylor Mackenzie Fisher, Alexsandro J. dos Santos and Sergi Garcia-Segura
Catalysts 2024, 14(5), 314; https://doi.org/10.3390/catal14050314 - 10 May 2024
Abstract
This study explores the use of the iron-containing metal–organic framework (MOF), Basolite®F300, as a heterogeneous catalyst for electrochemically-driven Fenton processes. Electrochemical advanced oxidation processes (EAOPs) have shown promise on the abatement of recalcitrant organic pollutants such as pharmaceuticals. Tetracyclines (TC) are
[...] Read more.
This study explores the use of the iron-containing metal–organic framework (MOF), Basolite®F300, as a heterogeneous catalyst for electrochemically-driven Fenton processes. Electrochemical advanced oxidation processes (EAOPs) have shown promise on the abatement of recalcitrant organic pollutants such as pharmaceuticals. Tetracyclines (TC) are a frequently used class of antibiotics that are now polluting surface water and groundwater sources worldwide. Acknowledging the fast capability of EAOPs to treat persistent pharmaceutical pollutants, we propose an electrochemical Fenton treatment process that is catalyzed by the use of a commercially available MOF material to degrade TC. The efficiency of H2O2 generation in the IrO2/carbon felt setup is highlighted. However, electrochemical oxidation with H2O2 production (ECO-H2O2) alone is not enough to achieve complete TC removal, attributed to the formation of weak oxidant species. Incorporating Basolite®F300 in the heterogeneous electro-Fenton (HEF) process results in complete TC removal within 40 min, showcasing its efficacy. Additionally, this study explores the effect of varying MOF concentrations, indicating optimal removal rates at 100 mg L−1 due to a balance of kinetics and limitation of active sites of the catalysts. Furthermore, the impact of the applied current on TC removal is investigated, revealing a proportional relationship between current and removal rates. The analysis of energy efficiency emphasizes 50 mA as the optimal current, however, balancing removal efficiency with electrical energy consumption. This work highlights the potential of Basolite®F300 as an effective catalyst in the HEF process for pollutant abatement, providing valuable insights into optimizing electrified water treatment applications with MOF nanomaterials to treat organic pollutants.
Full article
(This article belongs to the Section Environmental Catalysis)
►▼
Show Figures
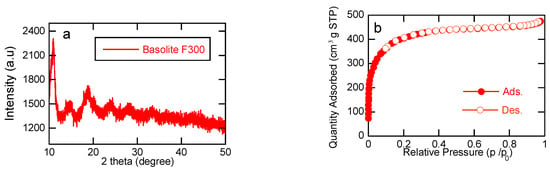
Figure 1
Open AccessArticle
Perovskite Oxide Catalysts for Enhanced CO2 Reduction: Embroidering Surface Decoration with Ni and Cu Nanoparticles
by
Andrea Osti, Lorenzo Rizzato, Jonathan Cavazzani, Ambra Meneghello and Antonella Glisenti
Catalysts 2024, 14(5), 313; https://doi.org/10.3390/catal14050313 - 10 May 2024
Abstract
The imperative reduction of carbon dioxide into valuable fuels stands as a crucial step in the transition towards a more sustainable energy system. Perovskite oxides, with their high compositional and property adjustability, emerge as promising catalysts for this purpose, whether employed independently or
[...] Read more.
The imperative reduction of carbon dioxide into valuable fuels stands as a crucial step in the transition towards a more sustainable energy system. Perovskite oxides, with their high compositional and property adjustability, emerge as promising catalysts for this purpose, whether employed independently or as a supporting matrix for other active metals. In this study, an A-site-deficient La0.9FeO3 perovskite underwent surface decoration with Ni, Cu or Ni + Cu via a citric acid-templated wet impregnation method. Following extensive characterization through XRD, N2 physisorption, H2-TPR, SEM-EDX, HAADF STEM-EDX mapping, CO2-TPD and XPS, the prepared powders underwent reduction under diluted H2 to yield metallic nanoparticles (NPs). The prepared catalysts were then evaluated for CO2 reduction in a CO2/H2 = 1/4 mixture. The deposition of Ni or Cu NPs on the perovskite support significantly enhanced the conversion of CO2, achieving a 50% conversion rate at 500 °C, albeit resulting in only CO as the final product. Notably, the catalyst featuring Ni-Cu co-deposition outperformed in the intermediate temperature range, exhibiting high selectivity for CH4 production around 350 °C. For this latter catalyst, a synergistic effect of the metal–support interaction was evidenced by H2-TPR and CO2-TPD experiments as well as a better nanoparticle dispersion. A remarkable stability in a 20 h time-span was also demonstrated for all catalysts, especially the one with Ni-Cu co-deposition.
Full article
(This article belongs to the Special Issue Advanced Research of Perovskite Materials as Catalysts)
►▼
Show Figures

Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Catalysts Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Batteries, Catalysts, Energies, Hydrogen, Sustainability
Preparation, Storage, and Transportation of Green Hydrogen and Multi-Scenario Application Technologies
Topic Editors: Weihua Cai, Chao Xu, Zhonghao Rao, Fuqiang Wang, Ming GaoDeadline: 30 June 2024
Topic in
Catalysts, Coatings, Crystals, Energies, Materials, Nanomaterials
Interfacial Bonding Design and Applications in Structural and Functional Materials
Topic Editors: Junlei Qi, Pengcheng Wang, Yaotian YanDeadline: 20 July 2024
Topic in
Energies, Catalysts, Fermentation, Processes, Waste
Valorizing Waste through Thermal and Biological Processes for Sustainable Energy Production
Topic Editors: Margarida Gonçalves, Cândida VilarinhoDeadline: 31 August 2024
Topic in
Cancers, Catalysts, Current Oncology, Plasma, Sci
Advances in Low-Temperature Plasma Cancer Therapy
Topic Editors: Michael Keidar, Li Lin, Dayun YanDeadline: 20 September 2024

Conferences
Special Issues
Special Issue in
Catalysts
Design and Synthesis of Nanostructured Catalysts, 2nd Edition
Guest Editor: Dinesh KumarDeadline: 25 May 2024
Special Issue in
Catalysts
Advances in Photocatalytic and Photothermal Catalytic CO2 Reduction
Guest Editor: Fernando FresnoDeadline: 6 June 2024
Special Issue in
Catalysts
Modern Catalytic Reactor: From Active Center to Application Tests, 2nd Edition
Guest Editors: Anna Gancarczyk, Agnieszka CiemięgaDeadline: 15 June 2024
Special Issue in
Catalysts
Visible Light Photocatalysis: Mechanisms and Applications
Guest Editors: Kunlei Wang, Zhishun Wei, Zuzanna BielanDeadline: 26 June 2024
Topical Collections
Topical Collection in
Catalysts
Photocatalytic Water Splitting
Collection Editors: Weilong Shi, Guigao Liu
Topical Collection in
Catalysts
Catalytic Conversion of Biomass to Bioenergy
Collection Editors: Sergio Nogales Delgado, Juan Félix González, Simona M. Coman






